Healing Halogens-Today's Knowledge Share
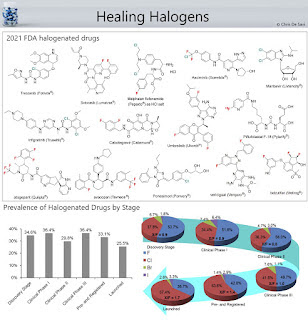
Today's Knowledge Share: Healing Halogens A significant number of drugs are halogenated. Typically, insertion of halogen atoms on hit or lead compounds is performed to occupy the binding site of molecular targets in order to improve the drug−target binding affinity and/or reduce metabolism. 14 out of the 50 molecules approved by the FDA in 2021 contain halogens. The introduction of F onto a chemical scaffold is able to infer changes that affect the physicochemical properties and the conformation of a molecule. Being the most electronegative element in the periodic table, F plays an important role in the modulation of pKa of neighboring functionalities. Substituting F for H on aromatic groups is also well known to improve metabolic stability. Lipophilicity is also affected by the addition of F onto aliphatic and aromatic scaffolds. The monofluorination or trifluoromethylation of saturated alkyl groups usually decreases the lipophilicity due to the strong electron-withdrawing capa



